Wieland Chase Explains Processes Leading to Improved Dezincification-Resistance
By Larry Muller, Senior Technical Advisor, Wieland Chase, LLC
Some technical papers and websites on brass corrosion repeat the same caution that dezincification will occur in alloys containing more than 15-percent zinc. A few will go beyond that simple statement to talk about efforts to solve this issue. The 15-percent zinc limit is generally the only thing that stays in the readers’ mind if they are trying to design a part that is in an environment that could lead to dezincification.
Reading this was a surprise to me after spending 28-plus years as a metallurgist working with a variety of copper and brass alloys. After all, our industry had sold billions of pounds of C36000, a 35+-percent zinc-leaded brass, to the potable water industry without corrosion problems outside of installation or grounding issues. If 15-percent zinc was the dividing line between a suitable brass for corrosion resistance and one prone to failure, where were the complaints, and why did the plumbing industry continue to use it?
The reason behind this 15-percent zinc content concern is lost to history. Older technical papers don’t provide a trail to the problems that caused someone to establish 15-percent zinc as an upper limit. It just gets repeated in formal and informal articles and papers on corrosion of brass without any attribution or investigation. The problem is that “15-percent zinc” isn’t the full story.
The history of brass used in water applications for the last 100 years shows that brass alloys with up to and over 35-percent zinc have been used successfully to the tune of multiple billions of parts in the United States and Europe. It didn’t happen without some effort, but it did happen, as the following examples show:
- The problem of dezincification in 70-percent copper/30-percent zinc brass tubes in steam-powered navy ship condensers was solved through an addition of an alloying element corrosion inhibitor (1).
- The problem of severe dezincification in potable water fittings was solved in Europe in 1980 with the introduction of a brass (CZ132/CW602N) using a high temperature anneal and addition of an alloying element corrosion inhibitor to leaded brass containing 36 percent zinc (2).
- In the United States, alloys C36000 and C37700 with about 36 percent and 39 percent zinc, respectively, were used successfully for decades without significant issues with dezincification.
- New lead-free brasses have been developed and commercialized with zinc contents greater than 15 percent, some using the single or double approach of alloying element and thermal processing to be dezincification-resistant.
Development of Dezincification-Resistant Brasses With Greater Than 15-Percent Zinc
It’s obvious then that brasses with zinc greater than 15 percent show no dezincification problems when using the metallurgical advancements of the last 100 years. The following provides explanations and data on what tools are used to make those brass alloys dezincification-resistant, what is meant by dezincification-resistant, and the tests that indicate they will provide long, functional lives.
What Does Dezincification-Resistant Mean and How Is It Tested?
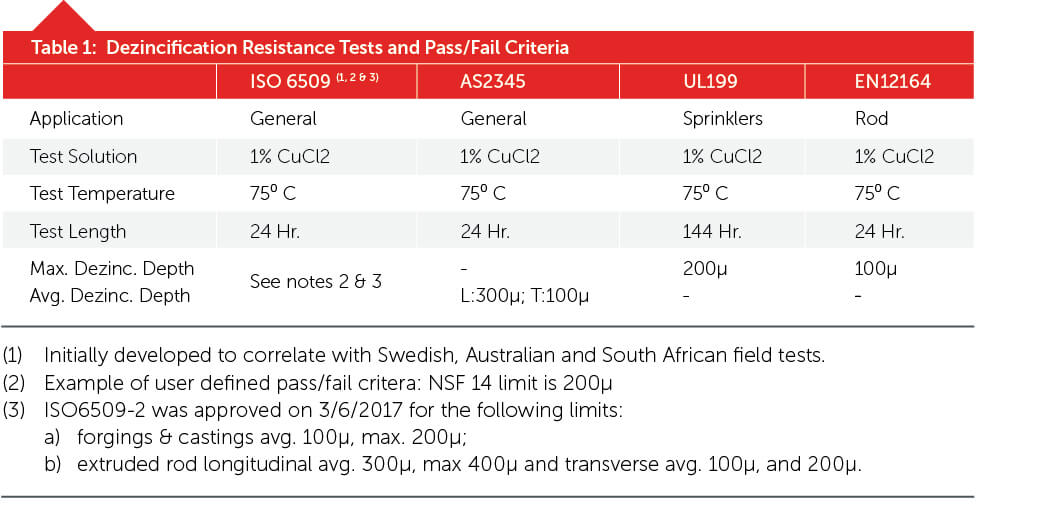
The old 15-percent zinc claim that brass “suffers dezincification” is vague. A brass part with zinc levels below 15 percent can show small levels of dezincification according to some published studies, and those with up to 35 percent can show no dezincification. Because the problem can affect alloys with up to 15-percent zinc, obviously some dezincification was considered acceptable. Recently, the brass world changed with the requirement to eliminate the use of leaded brass in potable water fittings (3, 4) and some high-profile PEX fitting dezincification failures in the Las Vegas area (5, 6). It became necessary to: 1) develop new lead-free alloys; 2) to define what “dezincification-resistant” meant quantitatively (7–11); and 3) have tests that would provide guidance on how long a part can be expected to last.
According to the various standards referenced in Table 1, a brass is considered dezincification resistant if it meets the requirements of the standards shown. There are other tests (Turner, Brandl) but the ISO6509 test has been the international standard since 1981. Independent studies (12–14) have shown this test successfully predicts long-term (greater than 30 years in corrosive drinking water) performance. An important point here is that this laboratory test uses extremely harsh conditions to accelerate the potential for corrosion that will take years or decades to develop. 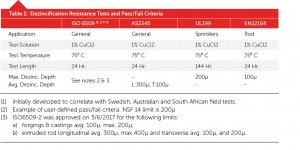
What Metallurgical Tools Are Used to Make Alloys Meet the “Dezincification-Resistant” Requirements?
There are three primary tools that make brass dezincification-resistant according to current requirements.
Minor alloying additions (0.20 percent maximum): Arsenic (As), Antimony (Sb), Phosphorus (P). These are the alloying element additions that have been researched and put into production over the last 100 years. There’s enough laboratory and real-life performance data to show that they are effective in preventing dezincification in brasses with zinc contents up to 36 percent (Figure 1). Above 36-percent zinc, neither these nor the major alloying elements shown can fully protect the brass because of a zinc-rich phase (beta phase) that forms during processing.
Major alloying additions (>0.25 percent): Nickel (Ni), Tin (Sn), Aluminum (Al). These alloying element additions have been in use longer than the minor alloying additions mentioned above. They are just as effective as the minor additions but have the same limitations of not being able to protect brasses with greater than 36-percent zinc because of the beta phase vulnerability to dezincification.
Thermal treatments such as high temperature annealing or forging. This method reduces or eliminates the beta phase in higher zinc brasses. By converting beta phase to alpha phase, it can now be protected by the minor or major alloying additions. The precaution here is that the cooling rate after these thermal treatments has to be slow enough to prevent beta phase from reforming. Fortunately, that process is well understood by the brass mills performing the operation.
What Data Show How These Metallurgical Techniques Improve Dezincification Resistance?
Figures 1, 2, and 3 show ISO6509 test results from an independent laboratory. Figure 1 shows the range of results from having no special metallurgical protection to alloying plus thermal treatments on alloys from 5-percent zinc to 42-percent zinc. At some point above 36-percent zinc, beta phase begins to form increasing amounts, so nothing can protect it as discussed above (Note: All test results are from Corrosion Testing Laboratories, Newark DE).
In Figure 2, you can see that the use of the two methods of the minor alloying addition and thermal treatment combine to improve dezincification resistance. However, they can only do so much before the beta phase problem negates their beneficial effects.
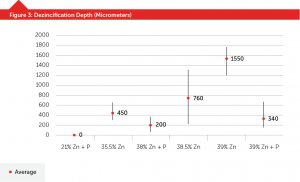
Figure 3 shows ISO6509 results for alloys with or without phosphorus and no thermal heat treatments. Except for the 21-percent zinc alloy, there’s a large fluctuation in results until the phosphorus addition is made. 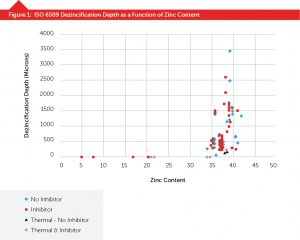
Improving Dezincification-Resistance
The data and information above show:
- the use of dezincification resistance alloying elements are an effective way to significantly improve dezincification resistance in brass with greater than 15-percent zinc;
- publications provide evidence that theses metallurgical technologies have been in use in production brass alloys for up to 100 years;
- any shortcomings of metallurgically advanced brasses with greater than 15-percent zinc would have been revealed by now; and
- this metallurgical technology has been replicated successfully among a number of “new” brasses commercialized to meet the 2014 national low lead requirement.
For more data and explanations, watch the webinar “The Basics of Dezincification”: https://wieland-chase.com/wp-content/uploads/2020/02/Dezincification-Web-Class-ver-2-12-2020.pdf
References
- Lucey, V. F. (1965, June). The mechanism of dezincification and the effect of arsenic I. British Corrosion Journal, 9–14.
- Copper Development Association. (1982). Dezincification-resistant brass. Information Sheet 36. https://copperalliance.org.uk/resources/36-dezincification-resistant-brass/
- California Assembly Bill no. 1953 (amended May 25, 2006). https://leginfo.legislature.ca.gov/faces/billNavClient.xhtml?bill_id=200520060AB1953
- Reduction of Lead in Drinking Water Act. (January 5, 2010). https://www.congress.gov/bill/111th-congress/senate-bill/3874
- Pope, J. (2008, November 4). $90 million faulty plumbing settlement gets initial approval. Las Vegas Sun.
- Sarver, E. (2010). Insights into non-uniform copper and brass corrosion in potable water systems [Doctoral dissertation, Virginia Tech]. https://vtechworks.lib.vt.edu/handle/10919/29474
- American National Standards Institute. (2019). Plastics piping system components and related materials (NSF/ANSI 14–2019).
- ISO. (2017). Corrosion of metals and alloys. Determination of dezincification resistance of copper alloys with zinc—Part 2: Assessment criteria (ISO6509-2: 2017).
- UL. (2001). Automatic Sprinklers for Fire Protection Service (UL199).
- Australian Standard. (2006; reconfirmed 2016). Dezincification resistance of copper alloys (AS 2345-2006).
- German Institute for Standardization-European Standards. (2016). Copper and copper alloys: rod for free machining purposes (DIN EN 12164: 2016-11). doi: https://dx.doi.org/10.31030/2535041
- Saver, Edwards, & Zhang. (2011). Revisiting dezincification performance for brass plumbing Devices. Materials Performance, 50(5), 70–75.
- Holm, R., Sundberg, R., & Mattson, E. (1982). Experiences with brass components for water installations in Sweden. Proceedings of the International Symposium on Corrosion of Copper and Copper alloys in Building, Tokyo, Japan, 230–238.
- Johnson. (1977, August). Evaluation of rapid tests for dezincification-resistant brass. BNF Metals Technology Center Research Report No. A 1921.


